



Non-Invasive Quantitative Liver Diagnostic Device for Reliable Stiffness and Steatosis Assessment
Quick Info.
- SKU NO.: UMY-US-122
- Device Classification: Class Ⅱ
- Warranty: 1 Year
- Power Source: Electric
- Transport Package: Carton or Wooden Box
- Origin: China
- Material: Metal, Plastic
- After-Sale Service: Online Technical Support
- Production Capacity: 1000 Sets/Year
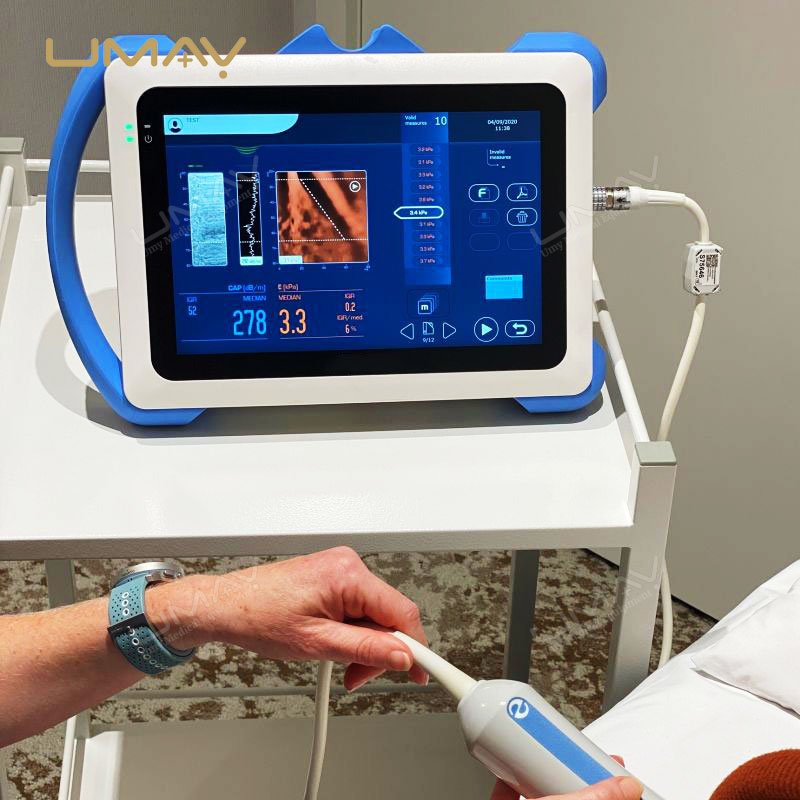
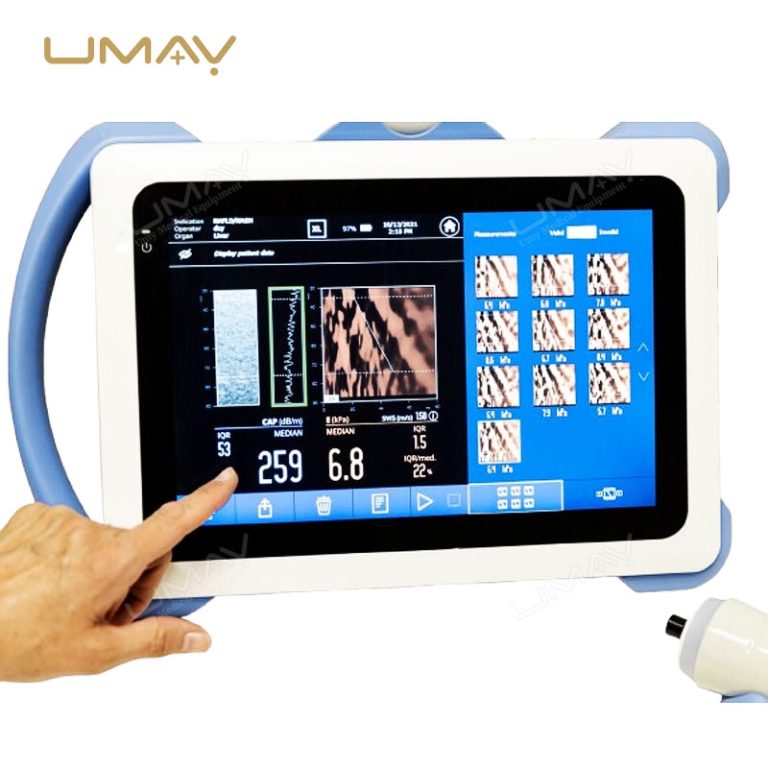
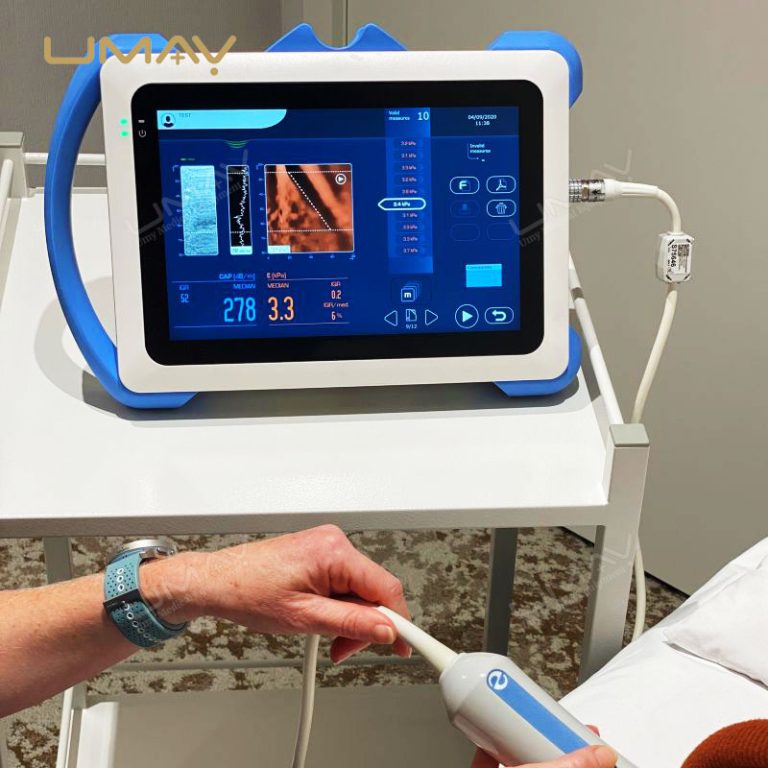
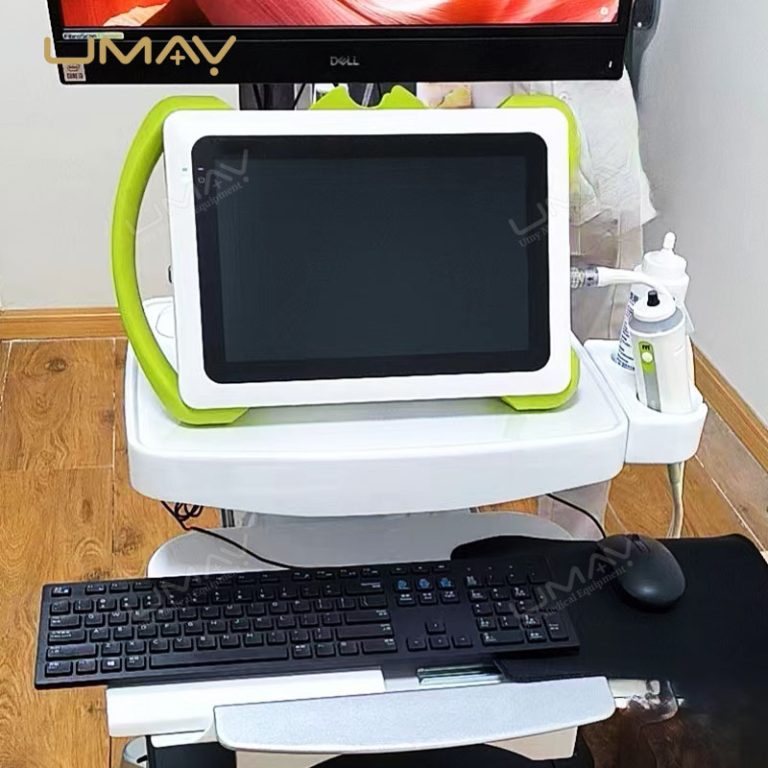
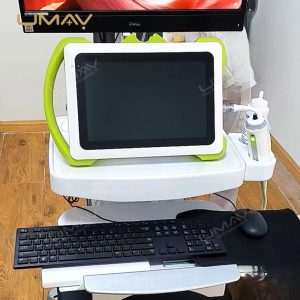
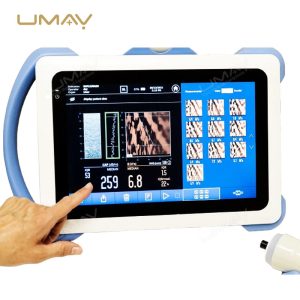
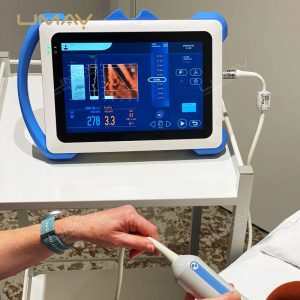
This non-invasive liver diagnostic device utilizes Vibration-Controlled Transient Elastography and Controlled Attenuation Parameter technology to provide precise measurements of liver stiffness and fat content. The device effectively evaluates liver conditions such as chronic hepatitis, non-alcoholic fatty liver disease (NAFLD), alcoholic liver disease (ALD), and drug-induced liver injury (DILI). With immediate results and high reproducibility, it’s suitable for routine screening, disease monitoring, and post-liver transplant follow-up. Supported by extensive clinical validation, this portable system ensures accurate diagnostics to inform treatment decisions confidently.
The Specific Parameters
Portability: Portable, lightweight design for patient screening and follow-up anywhere
Measurement Volume: Measures over 3 cm³ liver volume (at least 100 times larger than biopsy samples)
Results Display: Stiffness results expressed as E (kPa)
Liver Steatosis Measurement: Quantitative assessment via Controlled Attenuation Parameter
Probe Options: M+ and XL+ probes available
Clinical Applications: Chronic Hepatitis (B & C), HCV-HIV co-infection, ALD, liver cancer, MAFLD, biliary diseases, liver fibrosis, drug-induced injury, large-scale population screening
Advantages: Non-invasive, immediate results, clinically validated by over 3,500 peer-reviewed studies